Vaccines
Viral vaccine
- From 1962 to 2005, a whole-virus, inactivated trivalent influenza vaccine was produced.
- In 2005, the reconstruction of the facility according to cGMP standards began.
- In 2009, the Torlak Institute became part of the WHO Global Action Plan.
- 2020: TorVaxFlu® – Influenza vaccine (fragmented virus, inactivated).
The TorVaxFlu® vaccine is intended for protection against influenza in adults aged 18 and older.

Bacterial vaccines
Monovalent vaccines
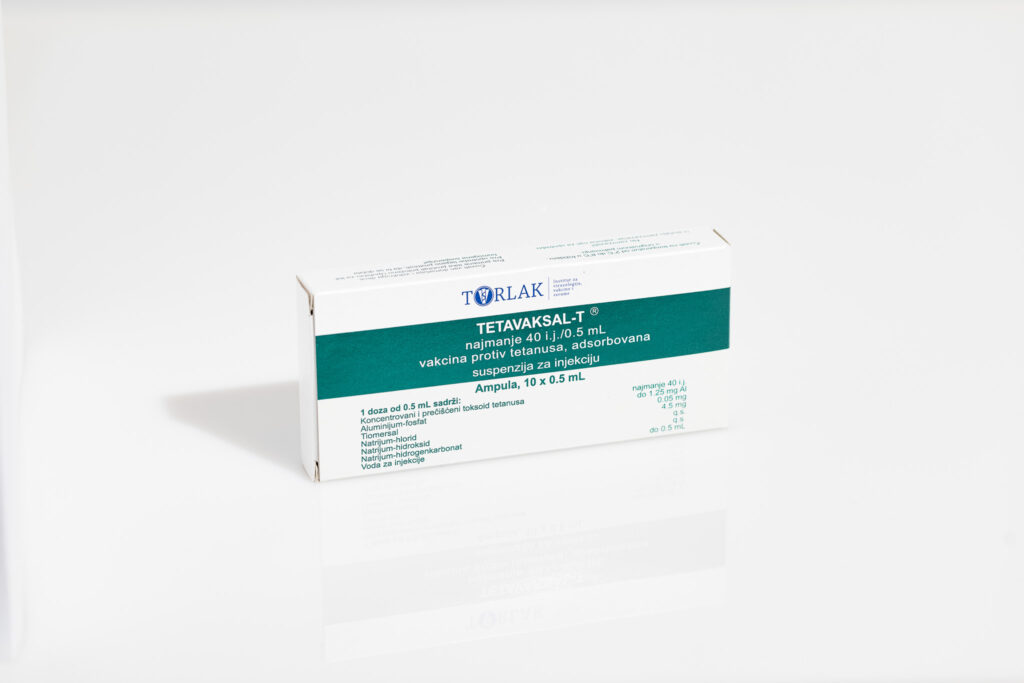
TETAVAXAL-T® – Tetanus vaccine, adsorbed
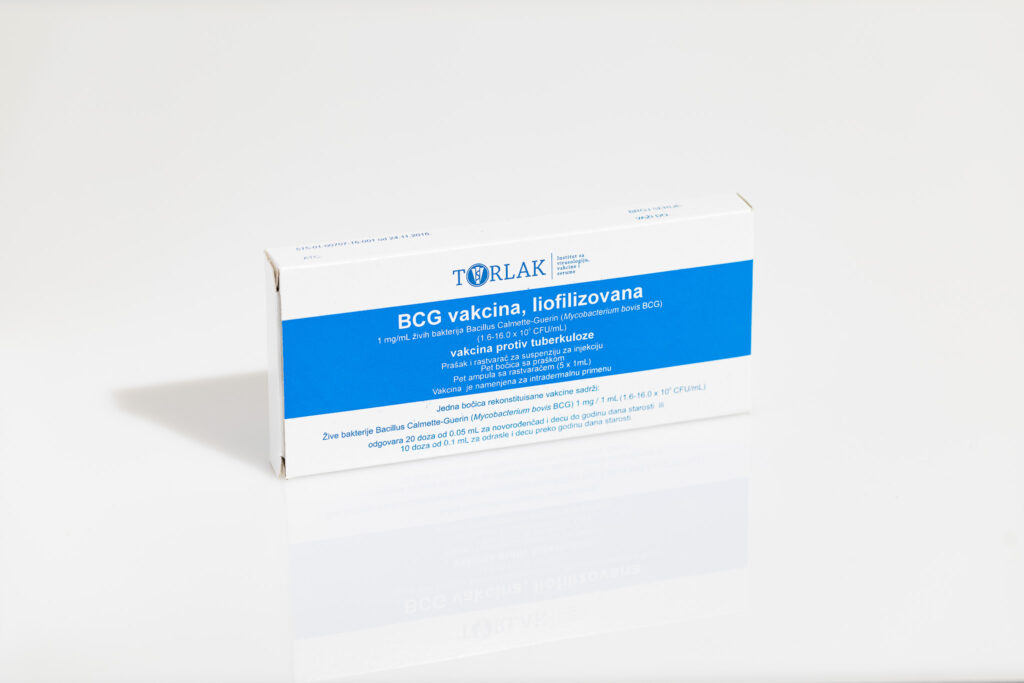
BCG vaccine, lyophilized – Tuberculosis vaccine
Polyvalent vaccines
DITEVAXAL-T® – Tetanus and Diphtheria vaccine, adsorbed

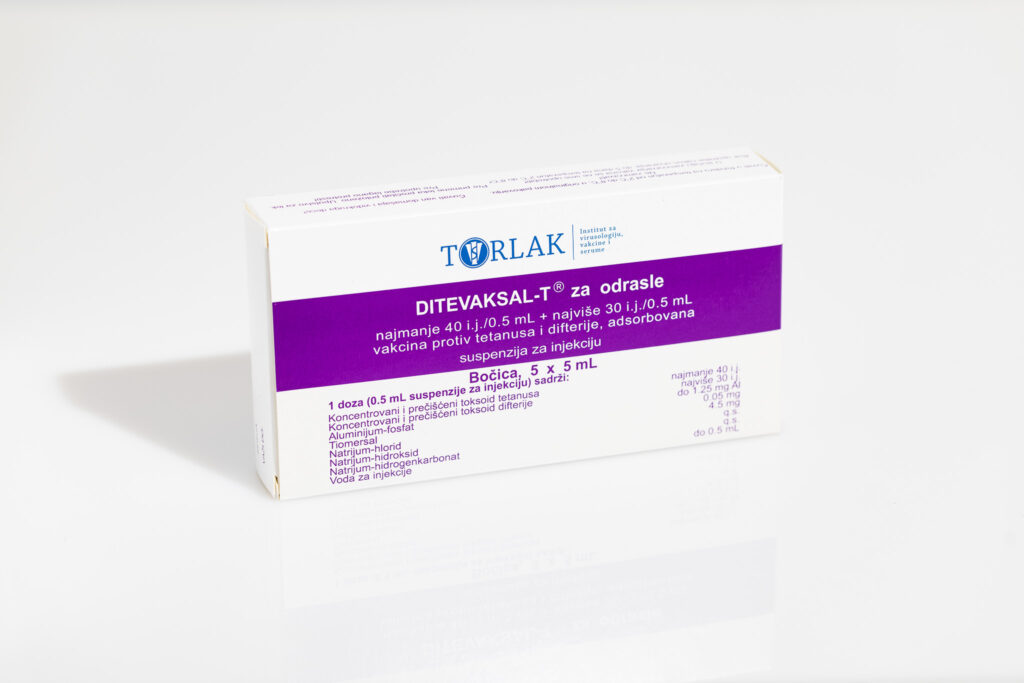
DITEVAXAL-T® for adults – Tetanus and Diphtheria vaccine, adsorbed
ALDIPETE-T® – Pertussis, Diphtheria, and Tetanus vaccine, adsorbed

